Drug Development Services
High quality and Speedy Full-Services for Drug Development
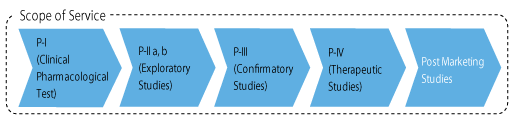
[Supported-Areas]
- Nationwide-Study
- Asian and Global-Study
[Major Services]
- Clinical Pharmacological Test: Phase I
- Monitoring
- Data Management
- Patient Registration Center
- Statistical Analysis
- Medical Writing
- Pharmacovigiliance
- Consultation, Regulatory and Others
[Clinical Pharmacological Test: Phase I]
- We are working closely with SEHMA CO., LTD. in providing high quality PK/PD data, as well as supporting services on Statistical Analytic Planning, Data Management, and Monitoring for our clients.
- SEHMA CO., LTD.
-Clinical Trial Liaison Office
-Preliminary Investigation
-Central IRB Management/Audit
-Clinical Trial Support Database Management
-Volunteer members' enrollment/registration
http://sehma.co.jp/index.htm
[Monitoring]
- Review of Medical Institutions and Investigators
- Explanation of Protocol to Investigators on GCP compliance
- Clinical Trial Plan Notification, Change Notification and Premature Termination/Completion Notification
- Request Clinical Trials to Medical Institutions and Process Site Contracts
- Response to IRB Review Results
- Contracts Finalization
- Arranging Startup Meetings
- Supply and Collection of Investigational Products
[Data Management]
- Design of CRF/DM
- Design of Database / Design of an Entry Screen
- Specifications of Logical/Manual Checks
- Development of Data Management System and System Validation
- Entry / Correction of Data / Coding
- Performance of Logical Checks and Manual Checks
- Development of DCF(CLF)
- Development of Materials for Case Review Meetings
- Database Lock
- Analysis Data-Set
[Patient Registration Center]
- Receipt of Subjects by Telephone / Fax / Web (Database Controlled Centrally)
- Computer-aided Contracting with Medical Institutions and Registration Physicians
- Balancing of Group Based on Prognosis Factors(Dynamic Assignment)
- Assignment of Investigational Products for Use
- Report of Entry Omissions and Discrepancies
- Eligibility Assessment in Accordance with Protocol
- Management of the Status of Enrollment
[Statistical Analysis]
- Protocol Development (Analysis-Related Portions, Sample Size Design,etc.)
- Development of a Statistical Analysis Plan
- Analysis Program, Output of the Results and Performance of Validation
- Interim Analysis
- Preparation of an Analysis at a Key-Opening Meeting
- Statistical Analysis Report
- Clinical Study Report (Analysis-Related Portions)
- Analysis for Development of an Application Dossier (in response to CTD)
- Consulting for Analysis Activities of Clinical Trials
[Medical Writing]
- Adverse Drug Reaction Reports, Clinical Study Reports
- Documents for Clinical Trials
- Post-Marketing Clinical Studies,
- Clinical Trial Plan Notifications,
- Protocol
- Informed Consent Written Information,
- Case Report Forms
- Investigator's Brochures
- Approval Applications and Reexamination Applications (Application Dossiers in Response to CTD, Reexamination Application Dossiers)
- Thesis Papers, Articles, etc
- Orphan Drug Designation Application Forms
- Preparation or Revision of Interview Forms
- Translation of reports (English and Japanese)
[Pharmacovigiliance]
- Preparation/ Regulatory Submission (Domestic and Overseas):
- Primary Assessment/ Translation Services(English and Japanese)
- Support for Instructions to Monitors and MRs
- Consultation for General Pharmacovigilance and related activities
- Consultation by Contract Specialist Physicians for Adverse Events
-CIOMS/MedWatch Forms
-Research Reports
-Adverse Drug Reaction
-Infection Case Reports
-Measure Reports
-Defect Report
-Various reports/documents for internal Safety Committees, HQ and Overseas Affiliates, etc.
-Clinical Trial/ Post-Marketing Safety Information -Overseas CIOMS/MedWatch Form
[Consultation, Regulatory and Others]
- Planning of Development Strategy, Clinical Trial Plans, and Various Reviews
- Consultation for Licensing in from Overseas and for Clinical Trials in Foreign Countries
- General/regulatory Consultation for Development of Drugs and Medical Devices
- Support for Studies in Humans on Health Food and Health Supplements
- Support for Investigator-Initiated Clinical Trials, Large-Scale Epidemiological Studies, etc.
- Support for Bioequivalence Studies on Generic Drugs and Applications
- Preparing and registration of Master File




